FACTORY
YILI Steel Structure
- Cost saving and transportation convenient, custom design available.
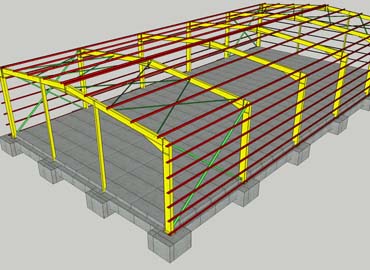
- The main beam and column are made of quality steel.
- Easy assembly and dis-assembly several times without damage.
- Good environmental protection effect. Steel structure construction greatly reduces sand, stone and dosage of the ash. Most of the used materials are recyclable or degradable, little rubbish produced.
- Own the hot dip galvanize zinc pool ourselves, pool size: 10.5x1.8x2.4m(LXWXD).
- Near the Qingdao port, only need 1.5hours by car,the land transport cost cheaper.
- Near the Qingdao International port, only need 0.5 hours by car, the client can visit us easily.
yili steel building
Steel Buildings
MULTI-FLOOR STEEL BUILDING
The multi-storied steel structures building offer ample flexibility in design and aesthetics for commercial metal buildings
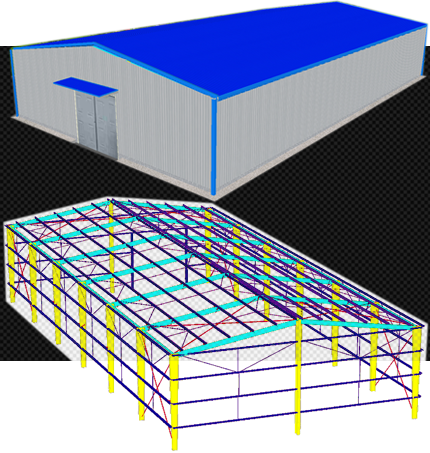
STEEL STRUCTURE BUILDING
A steel structure building is a metal structure fabricated with steel for internal support and for exterior cladding
STEEL STRUCTURE SYSTEM
The steel structure system shows the specification of the portal frame structure, bracing system, commercial mezzanine system